- Home
- Products
- Elementary
- Boride Powder
- 3D Printing Powder
- Sulfide Powder
- Oxide Powder
- Carbide powder
- Nitride Powder
- Silicide Powder
- Hydride Powder
- Telluride Powder
- Selenide Powder
- Stearic Acid Series
- Phosphide Powder
- Nanoparticles
- Metal Alloy
- MAX Phase
- Lithium Battery Anode
- Surfactant
- Molecular sieves
- Concrete Admixtures
- News
- Answers
- Contact
- About
News
- 1
- 1
The properties and uses of boron carbide and its synthesis process
If you are looking for high-quality products, please feel free to contact us and send an inquiry, email: brad@ihpa.net
Boron carbide (graph a) is a kind of black crystal with metallic luster, commonly known as black diamond. It is a powdery product made from boric acid as the main raw material, adding carbonaceous materials such as petroleum coke, and smelting and crushing in high-temperature solid state. It is similar to diamond, cubic nitride Boron is also a superhard material.
Boron carbide is insoluble in water and natural solvents, and has strong chemical stability, which is immune to acid and alkali rust, and does not react with mostly all acid and alkali services. Boron carbide also has the following attributes: it has a big thermal neutron capture cross-section and solid neutron absorption ability, so it is called a neutron absorber; it has semiconductor homes, and so on.
Properties and uses of boron carbide
Boron carbide (B4C) is gray-black and is a very hard man-made material with a Mohs firmness of 9.3 and a microhardness of 5500 ~ 6700kg/mm2, 2nd only to ruby and cubic boron nitride. The crystal structure of boron carbide is hexagonal crystal. Density is 2.52g/cm3. The melting point is 2450°C. When the temperature is higher than 2800°C, it decomposes and volatilizes rapidly. Its linear expansion coefficient is 4.5×10-6/℃ (20~1000℃), its thermal conductivity is 121.42(100℃)W(m·K), 62.80(700℃)W/(m·K), and its resistivity It is 0.44(20℃)Ω·cm and 0.02(500℃)Ω·cm. Boron carbide is immune to acid and alkali deterioration and does not wet with a lot of liquified steels, and has extremely high chemical security.Boron carbide can stand up to the oxidation of air at 1000 ° C, yet it is quickly oxidized over 900 ° C in an oxidizing setting. Boron carbide powder has a really high grinding capacity, which is 50% more than that of silicon carbide and 1 to 2 times more than that of diamond. It is an excellent abrasive material and wear-resistant material.
The biggest use of boron carbide is as an abrasive and a raw material for making abrasive tools. It is fit for grinding, polishing, drilling and other processing of various carbide tools, molds, parts, components and gemstones. Using an appropriate amount of engine oil or water as lubricant, boron carbide can be made into abrasive and polishing pastes. Boron carbide can also be used as a raw material for manufacturing metal borides, boron alloys, boron steel, etc. Except for special needs, it can be used to manufacture boron carbide hot-pressed products for wear-resistant and high-temperature-resistant parts, such as nozzles, sealing rings, gyroscopes, petrochemical parts, and lightweight and high-strength parts in military engineering. Control rods of an atomic reactor.

Synthesis process of boron carbide
The industrial method of synthesizing boron carbide is to reduce boron anhydride with excess carbon. The principle is:
2B2O3+7C→B4C+6CO
The synthesis reaction can be carried out in a resistance furnace or an electric arc furnace. A better method is to prepare boron carbide by heating a mixture of boron anhydride and carbon in a resistance furnace below the decomposition temperature of boron carbide. The boron carbide synthesized by this method contains very little free carbon and sometimes some free boron. During melting and synthesis in an electric arc furnace, since the temperature of the arc is quite high, boron carbide begins to decompose into a carbon-rich phase and boron at 2200°C. Boron will volatilize from the reaction space at high temperatures, so that the final reaction product contains a large amount of free carbon, the quality is poor. However, at present, domestic large-scale industrial production of boron carbide mostly uses arc fusion synthesis.
Three raw materials are used: boric acid (mass fraction greater than 92%), artificial graphite (mass fraction of fixed carbon greater than 95%), and stone coke (mass fraction of fixed carbon greater than 85%). According to the reaction formula, the proportioning synthesis reaction formula is:
4H3BO3+7C=B4C+6CO+6H2O
Among them, artificial graphite and petroleum coke each account for 50%. Considering factors such as high-temperature volatilization and oxidation in arc melting, the added amount of boric acid should be 2% higher than the calculated value, and the added amount of artificial graphite and petroleum coke should be 3% to 4% higher than the calculated value. After the three weighed raw materials are uniformly mixed in a ball mill, reduction and carbonization reactions are carried out in a single-phase two-electrode electric arc furnace or a three-phase electric arc furnace. The temperature of arc melting is controlled at 1700~2300°C. During the smelting process, it is best to feed materials in batches to block the arc light and achieve closed-arc smelting. After the smelting is completed, the melt is placed on carbon black or carbon electrodes to cool, and then the large pieces are smashed into 50mm-sized pieces with a hammer. Then the pieces are sorted according to their cross-sectional shape, and the unqualified pieces that are over-cooked and raw are removed.
According to experience, the cross-section of raw fired blocks is white or gray; the cross-sections of over-fired blocks are like graphite flakes or porous fused bodies; any steel-like dense cross-sections or metallurgical coke stripe-like cross-sections or sand-like cross-sections are For qualified blocks, the mass fraction of boron carbide is between 94% and 97%, and the mass fraction of free carbon is less than 1.5%. Then the qualified block is soaked in hot water and steamed, and then washed with water to remove the boric acid, boric anhydride and carbon substances adhering to it. The washed and dried large pieces are then coarsely crushed and passed through a 2.362mm sieve. Further coarse grinding and fine grinding can be carried out in a rotary ball mill or a vibrating ball mill. The coarse grinding time is shorter and the fine grinding time is longer. The iron added into the material during the ball milling process can be removed by leaching with hot sulfuric acid at 80°C. The amount of sulfuric acid added is 30% of the material. Pickling iron removal is carried out in a pickling tank. Add materials first, then add acid, then add 60' of water, stir with leaf slurry, and add steam to heat at the same time to increase the reaction speed. After pickling and soaking for 12 hours, dilute with water, ventilate and stir. After another 12 hours of incubation, remove the acid and wash with water until neutral. Finally, the purified material is sorted into granular products of different particle sizes using sedimentation fractions and serial water washing fractions. The settling semicolon is to divide the fine abrasive into fine powder of W35~W40; the series water washing semicolon is to divide the coarse abrasive into coarse particles of W40~120. If the coarse particles are further classified, the screening method can be used to divide the No. 120 particles into coarse particles of No. 100, No. 80, No. 70, No. 60 and coarser grades.
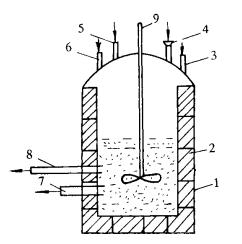
Schematic diagram of B4C pickling tank
(1-pickling tank shell; 2-acid-resistant tiles; 3-acid adding port; 4-feeding port; 5-steam inlet; 6-hot water inlet; 7-discharge port; 8-water outlet; 9-stirring device)
Product manufacturing
Due to the technical difficulty in manufacturing boron carbide special refractory products, industrial production has not yet been established. It is only manufactured in small quantities to meet the needs of some special occasions. At present, the manufacturing process of boron carbide products is not complete and mature. Products can be made by hot pressing firing method and normal temperature pressure firing method. Since the latter method is difficult to produce dense products, the former method is usually used. Hot pressing can be accomplished in a hot pressing furnace with a carbon tube heated under the protection of argon (Ar) gas. The heating speed is not strictly controlled, and it usually takes about 2 hours to reach the maximum temperature. The hot pressing temperature is 2050~2150℃, the hot pressing pressure is about 30MPa, and the maximum temperature is kept for 30 minutes. The final product has a density of 2.46~2.51g/cm3, a porosity of 0.4%~0.6%, a compressive strength of 2250MPa, and a flexural strength of 280MPa.

Supplier
TRUNNANO is a supplier of boron carbide materials with over 12 years experience in nano-building energy conservation and nanotechnology development. It accepts payment via Credit Card, T/T, West Union and Paypal. Trunnano will ship the goods to customers overseas through FedEx, DHL, by air, or by sea. If you are looking for high-quality concrete additives, please feel free to contact us and send an inquiry.
Inquiry us
PREVIOUS NEWS
building materials industry indispensable good material
NEXT NEWS
